Basic concepts of Thermal Engineering | Gate Mechanical Notes
- The pressure of the gas in terms of its mean kinetic energy per unit volume E is equal to 2E/3 and Kinetic Energy is always proportional to temperature.
- The super heated vapor acts as perfect gas and obeys all gas laws
- Absolute zero pressure will occur when there is no molecular momentum
- No liquid can exists at absolute zero pressure and absolute pressure can be attained at 0 Kelvin
- Behavior of gas can be fully determined by four gas laws
- For air ratio of specific heats is 1.4
- Mono-atomic gas has highest ratio of specific heat = 1.66As per Joule’s law internal energy is function of temperature.

- The same volume of all gases would represents their molecular weights
- Gases have two value of specific heat. One is at constant pressure and one is at constant volume
- The molecular weights of all the perfect gases occupy the same
- volume under same conditions of pressure and temperature.
- Work done in free expansion process is always zero
- Work done for rigid container having gas, is zero
- Heat and work are not properties of system
- Absolute value of internal energy cannot be found but only change in internal energy can be found
- On weight basis air contains 23 parts of oxygen
- Heat and work are path functions
- N.T.P means Normal Temperature Pressure
- Change in the enthalpy is the heat supplied during constant pressure
- Change in the internal energy is the heat supplied during constant volume
- For isothermal process, process index is unity
- The basis for measuring thermodynamic property of temperature is given by zeroth law of thermodynamics
- First law of thermodynamic gives concepts of internal energy
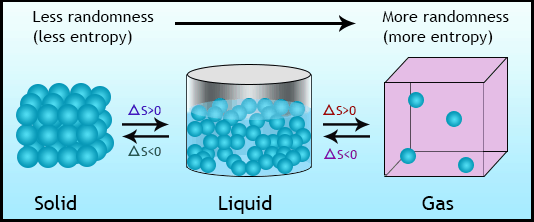
- Second law of thermodynamic gives concepts of entropy
- Third law of thermodynamic deals with concept of zero entropy at absolute zero temperature of 0 Kelvin.
- Throttling is the process during which cooling is produced
- 1 calorie = 4.186 Joule
- Integration of pdv is non flow work
- On volume basis air contains 21 parts of oxygen
- Molecular weight of gas x characteristics gas constant = value of universal gas constant
- For a perfect gas, internal energy and enthalpy are functions of temperature only.
More Resources /articles
Automobile Engineering Parts and System Notes , Article
Mechanical Subjectwise Basic Concept Notes ,Articles
New Mechanical Projects 2020 ( All Projects Post Index List )
Recent Posts
Mechanical Engineering is an essential discipline of engineering encompassing many specializations, with each contributing its unique aspect to the dynamic and inventive nature of this field. With...
The Ram Lalla idol, which is installed at Ayodhya's Ram temple has many significant religious symbols from Hinduism. All 10 incarnations of Lord Vishnu are engraved on the idol. Notably, Lord Ram is...