8 Basic Thermodynamic Processes – Basic Of Thermodynamics
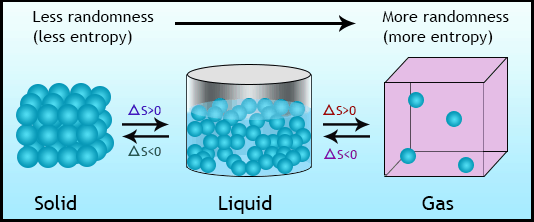
States of a thermodynamic system can be changed by interacting with its surrounding through work and heat. When this change occurs in a system, it is said that the system is undergoing a process.
A thermodynamic cycle is a sequence of different processes that begins and ends at the same thermodynamic state.
Some sample processes:
1. Isothermal process: temperature is constant T=C
2. Isobaric process: pressure is constant, P=C
3. Constant-volume process, v=C
4. Adiabatic process: no heat transfer, Q=0
5. Reversible process
6. Irreversible process
7. Quasistatic process

1. An isothermal process occurs at constant temperature. Since the internal energy of a gas is only a function of its temperature, ΔU = 0 for an isothermal process. For the isothermal expansion of an ideal gas we have W = nRT ln (V2/V1). W is positive if V2 > V1. Since ΔU = 0, the heat transferred to the gas is ΔQ = W.
2. An isobaric process is a process that occurs at constant pressure. We then have W = P(V2 – V1). If the pressure of an ideal gas is kept constant, then the temperature must increase as the gas expands. (PV/T = constant.) Heat must be added during the expansion process.
We define the enthalpy H of the system by the equation H = U + PV. The enthalpy is thus a physical property of the system. It has the dimensions of energy and the SI unit of enthalpy is Joule. For an isobaric process we write ΔU = ΔQ – ΔW = ΔQ – P(V2 – V1), or, rearranging the terms, ΔH = ΔQ.This expression, often used in chemistry, can be regarded as the isobaric form of the first law. ΔH = ΔQ only holds for isobaric processes. Chemical reactions (including biological ones) often take place at constant pressure, and then ΔQ is equal to the change in a physical property of the system.
3. An adiabatic process is a process during which no heat enters or leaves the system. We then have ΔU = -ΔW, i.e. ΔW equals the change in a physical property of the system. A physical property of the system depends only on the state of the system (P, V, T), not on the way the system was put into this state.
4. In practice there are two different ways to prevent the transfer of heat.
(a) Provide very good thermal insulation of the system.
(b) Complete the process in a very short time interval, so that there is no time for appreciable heat transfer. The combustion process inside a car engine is essentially adiabatic for this reason.
5. An isovolumetric or isometric process takes place at constant volume. Then W = 0 and ΔU = ΔQ. All the heat added to the system goes into increasing its internal energy.
6.Reversible process: The process in which the system and surroundings can be restored to the initial state from the final state without producing any changes in the thermodynamics properties of the universe is called a reversible process.
7. Irreversible process:
The irreversible process is also called the natural process because all the processes occurring in nature are irreversible processes.
8. Quasistatic process:
A quasistatic process is an idealized model of a thermodynamic process that happens infinitely slowly. It is important to note that no real process is quasistatic. In practice, such processes can only be approximated by performing them infinitesimally slowly. A quasistatic process often ensures that the system will go through a sequence of states that are infinitesimally close to equilibrium (so the system remains in quasistatic equilibrium), in which case the process is typically reversible.
Recent Posts
Mechanical Engineering is an essential discipline of engineering encompassing many specializations, with each contributing its unique aspect to the dynamic and inventive nature of this field. With...
The Ram Lalla idol, which is installed at Ayodhya's Ram temple has many significant religious symbols from Hinduism. All 10 incarnations of Lord Vishnu are engraved on the idol. Notably, Lord Ram is...